What Are Good Manufacturing Practices (GMP)?
Good manufacturing practices (GMP) are a system of procedures and controls used to make sure merchandise are constantly produced and meet outlined high quality requirements. It’s generally utilized in prescribed drugs, meals manufacturing and medical gadget manufacturing to stop contamination, errors and deviations throughout manufacturing. GMP establishes documented processes, skilled personnel necessities and managed environments to take care of product security and reliability.
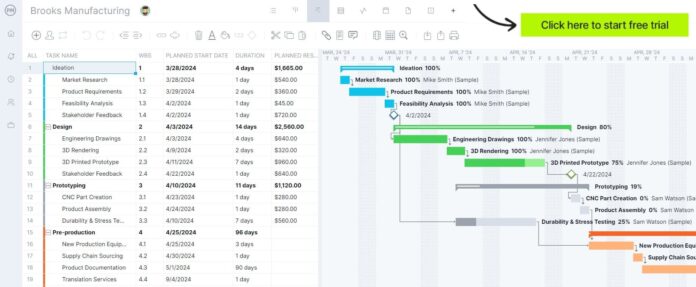
ProjectManager is an award-winning mission administration software program that’s outfitted with instruments that enable producers to plan, schedule and monitor their tasks from begin to end. Use Gantt charts to make manufacturing roadmaps, handle workflows with kanban boards, allocate assets with workload charts, observe prices with timesheets and monitor your tasks with real-time dashboards and reviews. Get began totally free at the moment.

Why Are Good Manufacturing Practices Essential?
Producers function in environments the place small course of failures can result in pricey defects, security dangers or regulatory motion. Implementing good manufacturing practices creates construction throughout operations, guaranteeing work is carried out constantly, documented correctly and monitored repeatedly. As manufacturing planning scales, this degree of management turns into important to take care of high quality, meet compliance necessities and keep away from disruptions that impression supply, popularity and profitability.
- Passing audits and inspections required for certifications or licenses
- Making certain traceability when investigating defects or conducting product remembers
- Sustaining constant product high quality throughout a number of manufacturing tasks
- Supporting international distribution by complying with worldwide GMP requirements
- Avoiding manufacturing shutdowns brought on by compliance violations
- Managing provider high quality and guaranteeing incoming supplies meet specs
- Defending model popularity when working in safety-critical industries
Advantages of Implementing Good Manufacturing Practices
Robust operational self-discipline creates measurable benefits past compliance. Adopting good manufacturing practices helps producers standardize workflows, scale back variability and enhance visibility throughout manufacturing. Over time, this results in fewer defects, higher useful resource utilization and extra predictable outcomes. As groups comply with structured processes and keep correct information, decision-making turns into quicker, dangers are simpler to handle and general operational efficiency improves.
- Improves product high quality by implementing standardized manufacturing processes
- Reduces rework and scrap brought on by course of inconsistencies
- Minimizes downtime by means of higher upkeep planning and course of management
- Enhances office security by implementing operational requirements
- Strengthens traceability throughout supplies, batches and completed items
- Reduces compliance dangers and potential regulatory penalties
- Improves provider coordination and materials high quality management
20 Good Manufacturing Practices (GMP)
Relatively than a single rule, good manufacturing practices (GMP) are utilized by means of a set of operational controls that govern how work is carried out, monitored and improved. Every space focuses on a selected a part of the manufacturing system, serving to producers keep consistency, scale back threat and meet compliance necessities as operations scale.
1. High quality Management (QC)
High quality management is the GMP that defines how supplies and merchandise are examined and verified in opposition to specs. It’s generally utilized in manufacturing to make sure merchandise meet high quality requirements earlier than launch. This contains sampling, testing procedures and laboratory knowledge administration.


Testing supplies the ultimate affirmation that merchandise meet required requirements earlier than reaching prospects. With out efficient high quality management, defects could go unnoticed till after distribution. Robust laboratory controls enhance accuracy, assist decision-making and guarantee solely compliant merchandise are launched, lowering threat and defending each the producer and the tip person.
2. Amenities & Premises
Amenities and premises is a GMP space that defines how manufacturing areas are designed, maintained and managed to assist secure manufacturing. It’s generally utilized in regulated environments to stop contamination, guarantee correct workflow and keep appropriate circumstances for manufacturing actions. This contains structure design, environmental controls and constructing upkeep requirements. Producers that wish to enhance the protection, effectivity and productiveness of their amenities can attempt strategies such because the gemba stroll or a 5s audit.


Manufacturing environments instantly affect product high quality, particularly when contamination or cross-contact is a priority. Poor structure design can create bottlenecks or enable supplies and personnel to intervene with one another. By sustaining managed, well-organized amenities, producers scale back operational dangers, enhance effectivity and make sure that processes could be executed with out compromising security or compliance necessities.
3. Gear & Utilities
This is without doubt one of the most necessary of all good manufacturing practices, because it governs the choice, operation and upkeep of equipment and assist techniques utilized in manufacturing. It’s generally used to make sure gear performs reliably and utilities like water, air and energy meet required requirements. This contains calibration, upkeep and efficiency monitoring procedures.


Dependable gear and steady utilities are important for constant manufacturing outcomes. When machines function exterior of specs or utilities fluctuate, product high quality could be affected with out instant detection. Implementing controls round gear and utilities helps forestall sudden failures, reduces downtime and ensures that manufacturing processes stay steady and repeatable over time.
4. Documentation & Recordkeeping
Relating to good manufacturing practices (GMP), documentation and recordkeeping set up how procedures, directions and manufacturing actions are documented and saved. It’s generally utilized in regulated manufacturing to make sure traceability, assist audits and confirm that processes have been adopted as supposed. This contains commonplace working procedures (SOPs), batch information and logs that seize operational knowledge.
Associated: 12 Key Manufacturing Paperwork (with Free Templates)
Clear documentation creates a dependable historical past of what was deliberate, executed and verified throughout manufacturing. When information are full and correct, groups can hint points, verify compliance and assist audits with out delays. Robust recordkeeping reduces confusion, improves accountability and supplies the proof wanted to reveal that manufacturing processes are managed and constantly executed.
5. Supplies Administration
Supplies administration controls how uncooked supplies and elements are obtained, saved, dealt with and tracked all through the manufacturing course of. It’s generally utilized in manufacturing to make sure supplies meet specs and stay appropriate to be used. This contains provider verification, stock monitoring and materials standing identification.
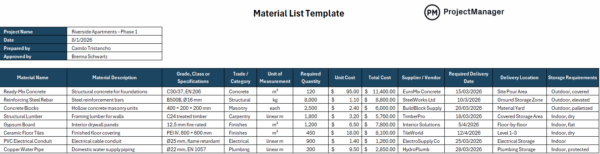

Incoming supplies instantly have an effect on last product high quality, so managing them correctly prevents defects from coming into manufacturing. With out clear controls, expired, contaminated or incorrect supplies can be utilized unintentionally. Establishing structured supplies administration improves traceability, reduces waste and ensures that solely accepted inputs are used, supporting constant and dependable manufacturing outcomes.
6. Manufacturing & Course of Controls
Manufacturing and course of controls outline how manufacturing operations are executed, monitored and managed. It’s generally used to make sure processes comply with outlined parameters and produce constant outcomes. This contains work directions, course of limits and in-process checks to take care of management throughout manufacturing actions.


Managed processes scale back variability and make outcomes extra predictable throughout manufacturing runs. When operators comply with standardized directions and monitor key parameters, points could be detected early earlier than affecting bigger batches. Robust course of controls enhance effectivity, reduce rework and assist producers keep constant product high quality whereas assembly regulatory expectations.
7. Packaging & Labeling
Packaging and labeling is the great manufacturing practices (GMP) space that governs how merchandise are packaged, labeled and ready for distribution. It’s generally used to make sure right identification, forestall mix-ups throughout order achievement and supply correct info to finish customers. This contains label verification, packaging controls and procedures for dealing with completed items.
Incorrect packaging or labeling can result in severe compliance points, product remembers or security dangers. By implementing strict controls, producers make sure that the proper product is packaged with the proper label each time. This reduces errors, protects shoppers and maintains belief whereas guaranteeing merchandise meet regulatory and market necessities.
8. Validation & Qualification
Validation and qualification guarantee processes, techniques and gear carry out constantly as supposed. It’s generally utilized in regulated manufacturing to substantiate that manufacturing strategies produce dependable and repeatable outcomes. This contains course of validation, gear qualification and system verification actions.
Unverified processes can introduce hidden dangers that have an effect on product high quality over time. By validating techniques and qualifying gear, producers achieve confidence that operations are steady and managed. This reduces uncertainty, prevents recurring points and helps constant manufacturing efficiency, particularly when scaling operations or introducing new processes or applied sciences.
9. Upkeep & Calibration
Upkeep and calibration is the method that ensures gear is serviced and adjusted to function inside outlined efficiency limits. It’s generally utilized in manufacturing to stop gear failures and keep measurement accuracy. This contains preventive upkeep schedules, calibration procedures and repair information to maintain techniques dependable and compliant.
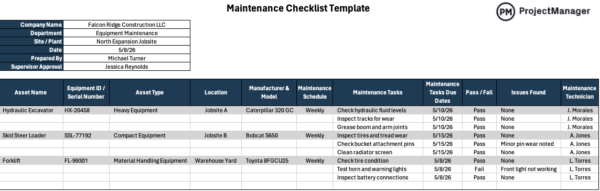

This is without doubt one of the most crucial good manufacturing practices of all of them as every bit of manufacturing gear tends to float over time, particularly underneath steady use in manufacturing environments. With out common upkeep and calibration, machines can produce inconsistent outputs or inaccurate measurements that impression product high quality. Establishing structured schedules reduces sudden downtime, improves reliability and ensures manufacturing processes stay steady and inside acceptable working circumstances.
10. Sanitation & Hygiene
Sanitation and hygiene is the sequence of tips, cleansing procedures and private cleanliness requirements required to stop contamination throughout manufacturing. It’s generally utilized in meals, pharmaceutical and beauty manufacturing to guard product integrity. This contains cleansing procedures, sanitation schedules and hygiene necessities for personnel and work areas.
Contamination dangers enhance rapidly when cleansing routines are inconsistent or poorly enforced. Even small lapses in hygiene can have an effect on complete manufacturing batches, resulting in waste or security considerations. By implementing strict sanitation controls, producers keep clear environments, scale back contamination dangers and guarantee merchandise are produced underneath circumstances that assist security and compliance.
11. Environmental Monitoring
Environmental monitoring is the method of monitoring circumstances corresponding to temperature, humidity, air high quality and microbial ranges in manufacturing environments. It’s generally utilized in managed manufacturing settings to make sure environmental elements stay inside acceptable limits. This contains routine measurements, testing protocols and recorded knowledge to confirm environmental stability.
Manufacturing environments can shift attributable to exterior elements or gear modifications, affecting product high quality with out instant visibility. Monitoring these circumstances permits groups to detect deviations early and take corrective motion earlier than points escalate. Constant environmental monitoring helps steady operations, protects delicate processes and ensures manufacturing circumstances stay appropriate for producing compliant merchandise.
12. Deviations, CAPA & Change Management
Deviations, CAPA and alter management is a GMP space that manages sudden points, corrective actions and managed modifications to processes or techniques. It’s generally used to research issues, tackle root causes and implement accepted modifications with out disrupting compliance. This contains deviation reviews, corrective motion plans and formal change approval workflows.
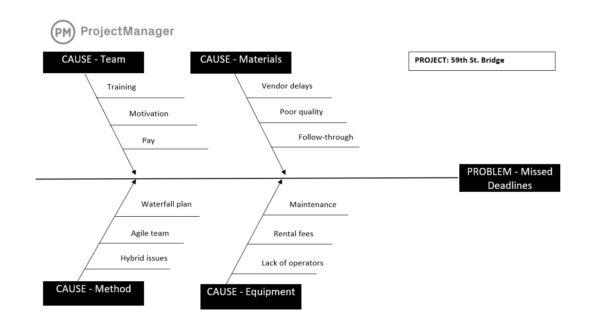

Issues are inevitable in any manufacturing setting, however how they’re dealt with determines long-term efficiency. With out structured controls, the identical points can repeat or modifications can introduce new dangers. Managing deviations and modifications systematically helps resolve points successfully, prevents recurrence and ensures all modifications are reviewed, accepted and documented earlier than implementation.
13. Product Recall & Returns
Product recall and returns are all the good manufacturing practices that set up how faulty or non-compliant merchandise are recognized, retrieved and managed after distribution. It’s generally utilized in manufacturing to take away unsafe merchandise from the market and hint affected batches. This contains recall procedures, return dealing with and traceability techniques to manage product motion.
As soon as merchandise attain prospects, any high quality subject turns into more durable to manage with no structured recall course of. Delays or gaps in traceability can enhance threat and regulatory publicity. Establishing clear recall and return procedures allows quick motion, limits impression, protects shoppers and helps producers reply successfully when product points are recognized out there.
14. Provider Relationship Administration
Provider relationship administration governs how exterior companions are chosen and monitored to make sure constant enter high quality. It’s generally utilized in manufacturing to confirm provider reliability and keep materials requirements. This contains provider qualification, efficiency analysis and accepted vendor lists.
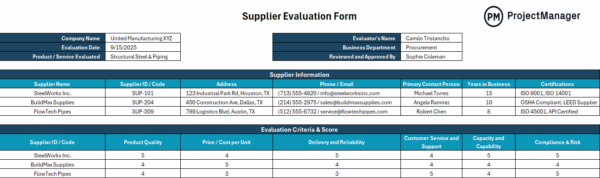

Manufacturing output relies upon closely on the standard of incoming supplies and outsourced providers. Weak provider oversight can introduce variability or defects earlier than manufacturing even begins. By managing distributors by means of structured evaluations and ongoing monitoring, producers scale back threat, enhance consistency and make sure that all inputs align with required specs and compliance expectations.
15. Distribution & Storage
Distribution and storage is a GMP space that controls how completed merchandise are saved, dealt with and transported to take care of high quality after manufacturing. It’s generally utilized in manufacturing to make sure merchandise stay inside outlined circumstances till they attain prospects. This contains storage controls, stock administration and transportation necessities.
Situations throughout storage and distribution can have an effect on product integrity, particularly for temperature-sensitive or regulated items. Poor dealing with or insufficient storage can result in degradation, contamination or non-compliance. By establishing clear controls over storage environments and transportation processes, producers shield product high quality, scale back losses and guarantee merchandise attain prospects in acceptable situation.
How ProjectManager Helps Manufacturing Companies
Maintaining manufacturing on schedule whereas managing assets, prices and altering priorities requires greater than spreadsheets or disconnected instruments. Manufacturing mission administration turns into simpler when groups can plan, observe and alter work in a single centralized system. ProjectManager brings collectively manufacturing scheduling, useful resource planning and efficiency monitoring so manufacturing groups can keep aligned and make quicker selections on the store flooring.
With interactive Gantt charts, manufacturing groups can construct detailed manufacturing schedules, map process dependencies, establish milestones and alter timelines as circumstances change. Useful resource administration instruments enable managers to assign labor, gear and supplies effectively, whereas monitoring useful resource availability and avoiding bottlenecks. Actual-time dashboards and reviews present prompt visibility into progress, prices and workload, serving to groups establish points early and keep management over manufacturing efficiency.
Groups may collaborate extra successfully utilizing kanban boards and process lists that observe work at each stage of the manufacturing course of. Thanks to those and different instruments and options, ProjectManager helps manufacturing companies enhance effectivity, scale back delays and ship constant outcomes. Watch the video beneath to study extra!
ProjectManager is award-winning software program for managing any mission. Our collaborative platform connects your groups and provides you entry to the manufacturing flooring regardless of the place you’re or what time it’s. Get began with ProjectManager at the moment totally free.